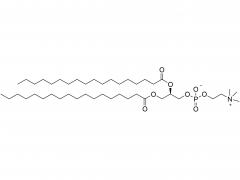
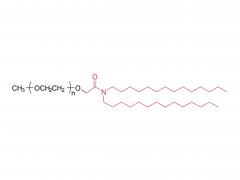
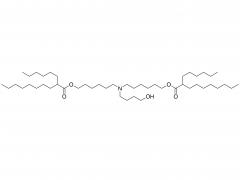
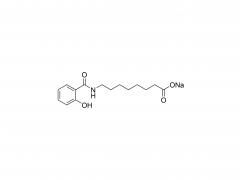
LNP delivery system
The major COVID-19 vaccine technology routes currently under development worldwide include inactivated vaccines, mRNA vaccines, adenovirus vaccines and recombinant protein vaccines. As a new vaccine technology in the market, mRNA vaccine is also one of the most important COVID-19 vaccines in the world at present. As a new technology, why can mRNA vaccine be widely administered around the world? One important reason is that it has a very high effective protection rate. The two available mRNA vaccines have an effective protection rate of more than 90%, and BioNTech's mRNA vaccine, produced in collaboration with Pfizer, has an effective protection rate of 95%.Since vaccination began, the daily positive rate in the United States has dropped from 20 percent to 1 to 2 percent.
MRNA transmits the genetic information for producing an antigen to the cells that make the protein. These cells then present the antigen to their surfaces, triggering the specific immune response needed. Eventually, when a virus invades, the immune system recognizes specific antigens and quickly and specifically attacks the virus to prevent infection. MRNA technology can not only be used as a preventive vaccine to prevent the spread of infectious diseases, but also as a therapeutic drug to treat some serious diseases, such as cancer and AIDS, due to its ability to spontaneously stimulate human immunity.
MRNA has large molecular weight, strong hydrophilicity and high biological activity, but its single chain structure makes it extremely unstable and easy to be degraded, and delivery through the membrane with negative charge on the surface is also difficult. MRNA must enter the cell to encode antibodies, and the enzyme degradation and cell membrane barrier in the process of entering the cell are the biggest challenges that affect its delivery efficiency and transfection efficiency. Special modification or package delivery systems are required to achieve intracellular expression of mRNA.
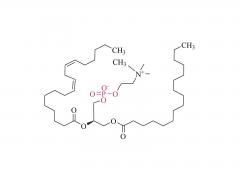
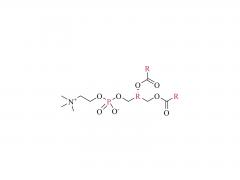
At present, Lipid nanoparticle (LNP) is commonly used as a carrier to deliver mRNA .Lipid nanoparticles mainly contain four components: ionizable lipids, neutral helper lipids, cholesterol, and PEGylated lipid. The neutral helper lipids are usually saturated phospholipids, which support the formation of the lamellar lipid bilayer and stabilize its structure arrangement. Cholesterol had strong membrane fusion, which promoted the intracellular uptake of mRNA and cytoplasmic entry. Pegylated lipids are located on the surface of lipid nanoparticles, improving their hydrophilicity and avoiding rapid removal by the immune system, preventing particle aggregation and increasing stability. The most critical excipients are ionizable cationic lipids, which are decisive factors in the efficiency of mRNA delivery and transfection.
Mechanism of LNP mRNA delivery: before entering cells, cationic lipids can realize electrostatic complexation with negatively charged mRNA molecules to form complex and improve the stability of mRNA molecules. When the mRNA/LNP complex arrived at the cell membrane, cationic lipids and the negatively charged cell membrane triggered the membrane fusion, which de-stabilized the cell membrane and promoted the entry and delivery of mRNA molecules. After complex internalized into cells, as lysosomes contain a variety of hydrolytic enzyme decomposition exogenous macromolecules, partial acid pH lower formation environment, make can be ionized lipid protons, LNP double-layer structure destruction, release the mRNA, according to the "central dogma" and in combination with the ribosome is responsible for the production of proteins, translated into viral proteins and antibodies, neutralize the virus.

In addition to the application of mRNA vaccine, LNP can also play an important role in gene editing therapy. Intellia Therapeutics and Regeneron recently announced that their co-development program, ntra-2001, a systemic CRISPR/Cas9 therapy, has achieved positive results in a phase I clinical trial. A single dose of NTLA-2001 resulted in an average 87% decrease in serum transthyroxine protein level (TTR), with a maximum reduction of 96% at day 28.As revealed in Intellia Therapeutics' patent, LNP contains amine lipids for encapsulation and in vivo escape, neutral and helper lipids for stabilization, and cloaking lipids. LNP raw materials used in CRISPR/Cas9 therapy include lipids such as DSPC, cholesterol, and PE2K-DMG, which are mostly similar to those used in LNP for mRNA vaccines.

MRNA /LNP COVID-19 vaccine excipients supply
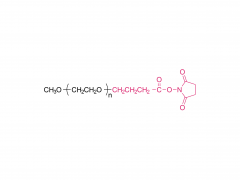
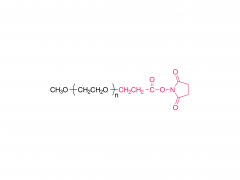
Lipids are the key of mRNA delivery system.Pfizer uses alC-3015 and ALC-0159, and Moderna uses SM-102 and PEG2000-DMG, respectively.
Lipids, as such high-end preparation excipients, have few suppliers to choose from, let alone domestic supply. In order to avoid the bottleneck of high-end excipients in the field of new technology such as mRNA/LNP, SINOPEG took the lead in the commercial production of LNP delivery materials in China. Currently, the company has several kinds of mRNA vaccine lipid excipients with independent intellectual property rights, and can be customized according to the customer's request. SINOPEG's various products (including customized structures) have been verified by several mRNA vaccine manufacturers, with complete quality research data.
The COVID-19 pandemic has greatly boosted the development of mRNA vaccines, while also leading to a shortage of LNP delivery vectors. At this critical moment, based on years of experience accumulation and strong technical backing, SINOPEG timely filled the supply gap of LNP delivery vector for global mRNA vaccine manufacturers, and realized the commercial production of mainstream lipid of mRNA vaccine in a short period of time to supply domestic and foreign enterprises. And can provide a number of independent intellectual property rights of lipids, to avoid European and American patent barriers, the development of a number of new lipid excipients successfully through downstream verification; At the same time to undertake customized development needs of customers, to solve their GMP production and declaration and other problems.
As a supplier of LNP delivery system excipients at home and abroad, the company leads the industry in the quality and industrialization of polyethylene glycol modified lipids and cationic lipids. And because the company is a leading global polyethylene glycol (peg) and its derivatives manufacturer, can provide from polyethylene glycol (peg) polymerization to polyethylene glycol modified lipid one-stop solution, since the outbreak in addition to continue to market supply mRNA vaccine lipid material, also for other lipid accessories manufacturers at home and abroad to provide high-quality polyethylene glycol and polyethylene glycol derivatives products, Such as MPEG-OH, MPEG-CM, MPEG-CH2CH2CH2-NH2, and MPEG-epoxide. SINOPEG is expected to play an increasingly important role in mRNA vaccines. For further information, please contact 0592-7761068 or sales@sinopeg.com.
ABOUT SINOPEG
XIAMEN SINOPEG BIOTECH CO., LTD. is a professional research and development, production and sales as one of the high-tech companies, mainly committed to drug delivery (DDS) and new medical materials business. The company was founded in 2011, has strong technical reserves and high-level team, research and development and production of high-quality polyethylene glycol, polyethylene glycol derivatives, block copolymers, blood glucose control drug modifiers, lipid excipients, ADC and ProTAC linker, etc.
Thanks to high quality products and efficient service, SINOPEG's products have been successfully used in PEGG-modified proteins, long-acting modification of peptides, mRNA vaccines and high-end third-class medical devices. The coverage of its main business in the domestic market has exceeded 60%, and more than ten of the top 20 domestic pharmaceutical enterprises have established long-term cooperation with the company. And successfully into North America, Europe (including Russia), South America and other Asia-Pacific regions. With long-term technical accumulation and highly recognized market support, SINOPEG has successfully entered the supply chain of mRNA vaccine excients and become a qualified supplier of many well-known COVID-19 mRNA vaccine manufacturers at home and abroad.
References
[1] Wei T , Cheng Q , Min Y L , et al. Systemic nanoparticle delivery of CRISPR-Cas9 ribonucleoproteins for effective tissue specific genome editing[J]. Nature Communications, 2020, 11(1).
[2] Buschmann M D , Carrasco M J , Alishetty S , et al. Nanomaterial Delivery Systems for mRNA Vaccines. 2021.